 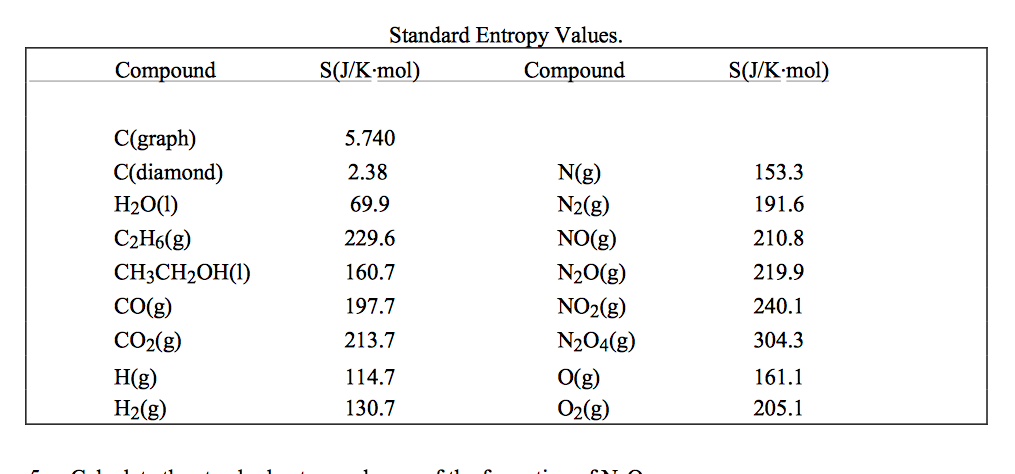
Referencing the enthalpy to h 25 slightly distorts the geometry of the diagram because h 25 is a function of composition. The y-axis is ( h − h 25) where h 25 is the molar enthalpy of the system in equilibrium at 25☌. 7.12B is the enthalpy-composition phase diagram corresponding to the T-composition diagram of Fig. 
The enthalpy axis is referred to the enthalpy of the system at equilibrium at 25☌ ( Pelton, 2014).įig. Corresponding T-composition and enthalpy-composition phase diagrams of the Mg-Si system at P = 1 bar. In essence, the reaction spontaneity is a matter of probability: namely, the system displaces spontaneously toward a state of maximum randomness (or less order) because that the type of state is the most probable.įig. However, the dissociation of CaSO 4 in water is spontaneous and exothermic. For example, the table salt dissociation in water is spontaneous- and endothermic. The spontaneity of a reaction does not have anything to do with its endothermic or exothermic character. If during a reaction process, heat is released into the ambient, then the reaction is referred to as exothermic otherwise, if the reaction receives heat, it is denoted as endothermic. Conversely, in a nonspontaneous chemical reaction, the composition of species moves away from the equilibrium. In a spontaneous chemical reaction, the mixture of species displaces toward the equilibrium composition. Combustion of a fuel, once ignited, occurs without any other external influence therefore, it is a spontaneous process, and it occurs at a very high rate. Iron oxide (or rust) is a spontaneous reaction that occurs at a very slow rate in usual atmospheric conditions. The spontaneity of a reaction does not have anything to do with the reaction rate. From a thermodynamic viewpoint, a spontaneous reaction is one that occurs in accordance to the second law of thermodynamics, which states that, for a process to be possible, it must evolve such that the generated entropy is positive. A nonspontaneous process is defined as the reverse of a spontaneous process. The spontaneity feature of a chemical reaction is defined as follows: if a chemical reaction occurs without any external influence, then it is denoted as spontaneous. 
For the purpose of this chapter, we will define chemical equilibrium as a state when the concentration of reactants and products remains constant over time. The first is chemical equilibrium, also discussed in Chapter 1. In order to tackle with such issues, some elementary notions must be introduced. From a thermodynamic point of view, several aspects need to be analyzed, and several questions need to be answered: namely, is the reaction spontaneous? For example, is it endothermic or exothermic? Does the chemical equilibrium tend toward generating more products? Or how carefully does one set needs to initially the amounts of reactants in the mixture such that more products are formed at equilibrium conditions? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
